A new standard of care: the benefits of corneal cross-linking with a non-invasive procedure1,2
Epithelium-on oxygen-enriched corneal cross-linking using Epioxa
- Leaves the epithelium intact for improved patient comfort, a streamlined procedure, and fast recovery time1,2
- Reduces the risk of infection and other serious adverse events2
- Is suitable for corneas as thin as 325 microns1
- Allows you to treat KC early without waiting to confirm progression1
- Incorporates oxygen enrichment1
Boost Goggles® supply optimal oxygen levels to maximize corneal cross-linking efficiency - Uses high-power photoenhancement1
Optimizes oxygen consumption to minimize procedure time
Drops, oxygen, light, action!1
Epioxa treatment has streamlined corneal cross-linking therapy:
PART 3

Deliver UV-A light with elevated irradiance lamp†
- The system emits UV-A radiation at a wavelength of 365 nm at an irradiance of 30 mW/cm2 pulsed (1 second on, 1 second off) for 11 min, 6 sec (total energy density of 10 J/cm2)1,4
- Requires the use of a Treatment Activation Card4‡
- The O2n System does not interface with private or public networks, including the internet4§
*The corneal mucin layer must be removed with a sponge soaked with Epioxa before applying drops.1
†Flush corneal surface with balanced salt solution as necessary to maintain corneal hydration.1
‡Included in the Epioxa Kit.
§This eliminates the risk of remote cyber attacks.
Epioxa procedure
Watch the video to understand how the Epioxa procedure works
There are 2 types of corneal cross-linking, differentiated by their use of oxygen5
A new video is coming soon: stay tuned to see the 2 types of corneal cross-linking in action
-
Type I Process6

UV-A LIGHT PHOTOENHANCER Lower
EfficiencyType I mainly involves a photoenhancer and UV-A light, is slow, and less efficient
-
Type II Process6

UV-A LIGHT PHOTOENHANCER SUPPLEMENTAL OXYGEN
Higher
EfficiencyType II with sufficient oxygen is more efficient
Type II corneal cross-linking is more efficient than Type I due to supplemental oxygen5,6
Oxygen is vital to a corneal cross-linking environment, but is rapidly depleted under UV-A7,8
Stromal O2 Concentration Before, During, And After UV9
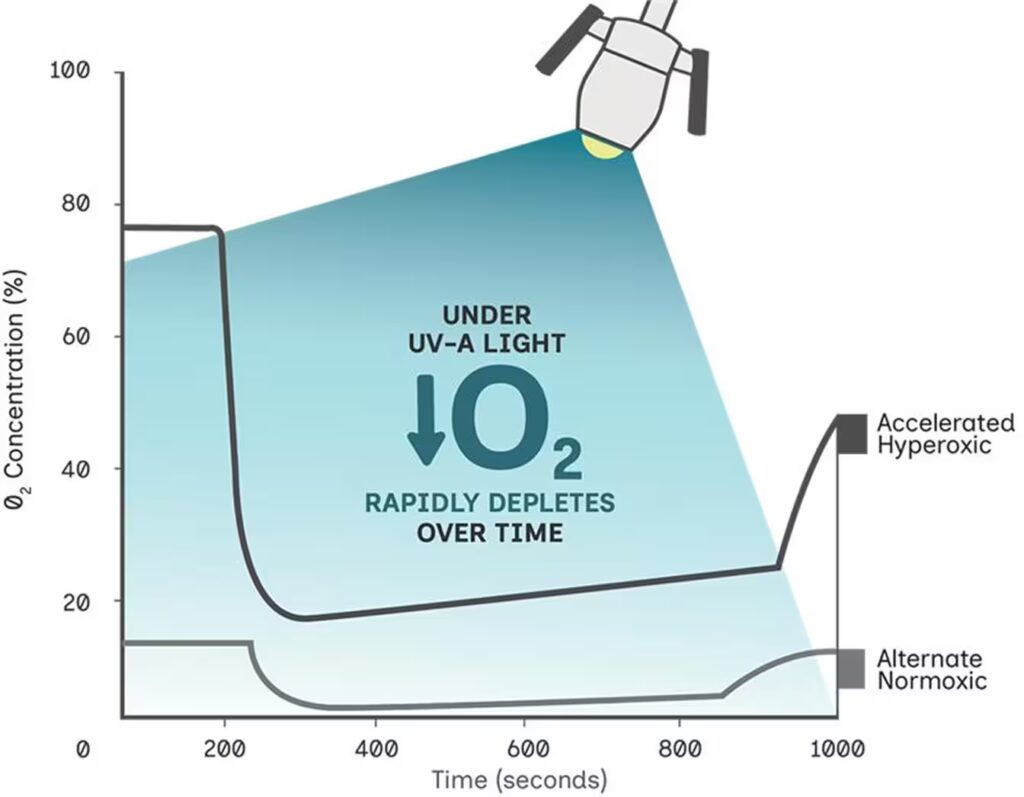
In oxygen-enriched environments, the Type II process dominates8
Without supplementation, oxygen is rapidly depleted under UV-A, the Type II process ceases, and only the slower, less-efficient Type I process occurs8
Supplemental oxygen is essential for epithelium-on corneal cross-linking9-11
Explore efficacy resultsKC=keratoconus; UV-A=ultraviolet-A.
References:
- EPIOXA HD and EPIOXA Prescribing Information. Glaukos Corporation; 2025.
- D’Oria F, et al. Corneal collagen cross‑linking epithelium‑on vs. epithelium‑off: a systematic review and meta‑analysis. Eye Vis (Lond). 2021;8(1):34.
- Data on File. Glaukos. Boost Goggles User Guide.
- Data on File. Glaukos. O2n System Operator’s Manual.
- Lin JT. Resolving the controversial issues and optimal new protocols for accelerated corneal cross-linking (CXL). Med Surg Ophthal Res. 2018;2(2):MSOR.000543. Available from: https://crimsonpublishers.com/msor/pdf/MSOR.000543.pdf
- Wu D, et al. Corneal cross-linking: the evolution of treatment for corneal diseases. Front Pharmacol. 2021;12:686630.
- Raiskup F, et al. Crosslinking with UV-A and riboflavin in progressive keratoconus: from laboratory to clinical practice – developments over 25 years. Prog Retin Eye Res. 2024;102:101276.
- Raiskup F, Spoerl E. Corneal crosslinking with riboflavin and ultraviolet A. I. principles. Ocul Surf. 2013;11(2):65-74.
- Hill J, et al. Optimization of oxygen dynamics, UV-A delivery, and drug formulation for accelerated epi-on corneal crosslinking. Curr Eye Res. 2020;45(4):450-458.
- Cronin B, et al. Oxygen-supplemented and topography-guided epithelium-on corneal crosslinking with pulsed irradiation for progressive keratoconus. J Cataract Refract Surg. 2024;50:209-216.
- Matthys A, et al. Transepithelial corneal cross-linking with supplemental oxygen in keratoconus: 1-year clinical results. J Refract Surg. 2021;37(1):42-48.
IMPORTANT SAFETY INFORMATION
Contraindications
EPIOXA™ HD and EPIOXA™ are contraindicated in patients with known hypersensitivity to benzalkonium chloride (BAC) or any ingredients in EPIOXA HD and EPIOXA. Epithelium-on corneal collagen cross-linking is contraindicated in aphakic and pseudophakic patients without a UV-blocking intraocular lens.
Warnings and Precautions
Corneal collagen cross-linking should be used with caution in patients with a history of herpetic keratitis due to the potential for reactivation of herpes keratitis.
Adverse Reactions
The most common adverse reaction was conjunctival hyperaemia (31%). Other adverse reactions, occurring in 5% to 25% of eyes included: corneal opacity (haze), photophobia, punctate keratitis, eye pain, eye irritation, increased lacrimation, corneal epithelium defect, eyelid oedema, corneal striae, visual acuity reduced, dry eye, and anterior chamber flare.
Dosage and Administration
EPIOXA HD and EPIOXA are for topical ophthalmic use. NOT for injection or intraocular use.
EPIOXA HD and EPIOXA are supplied in single-dose syringes. Discard opened syringes after use.
EPIOXA HD and EPIOXA are for use with the O2n System and Boost Goggles only.
Refer to the O2n System Operator’s Manual and Boost Goggles User Guide for device instructions.
INDICATIONS AND USAGE
EPIOXA™ HD (riboflavin 5’-phosphate ophthalmic solution) 0.239% and EPIOXA™ (riboflavin 5’-phosphate ophthalmic solution) 0.177% are photoenhancers indicated for use in epithelium-on corneal collagen cross-linking for the treatment of keratoconus in adults and pediatric patients aged 13 years and older, in conjunction with the O2n™ System and the Boost Goggles®.
Please see full Prescribing Information for EPIOXA HD and EPIOXA at www.Epioxa.com.
You are encouraged to report all side effects to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.
You may also call Glaukos at 1-888-404-1644.