Epioxa treatment is a game-changer that helps improve corneal integrity and function1
Epioxa significantly improved corneal structural stability to help maintain visual function1
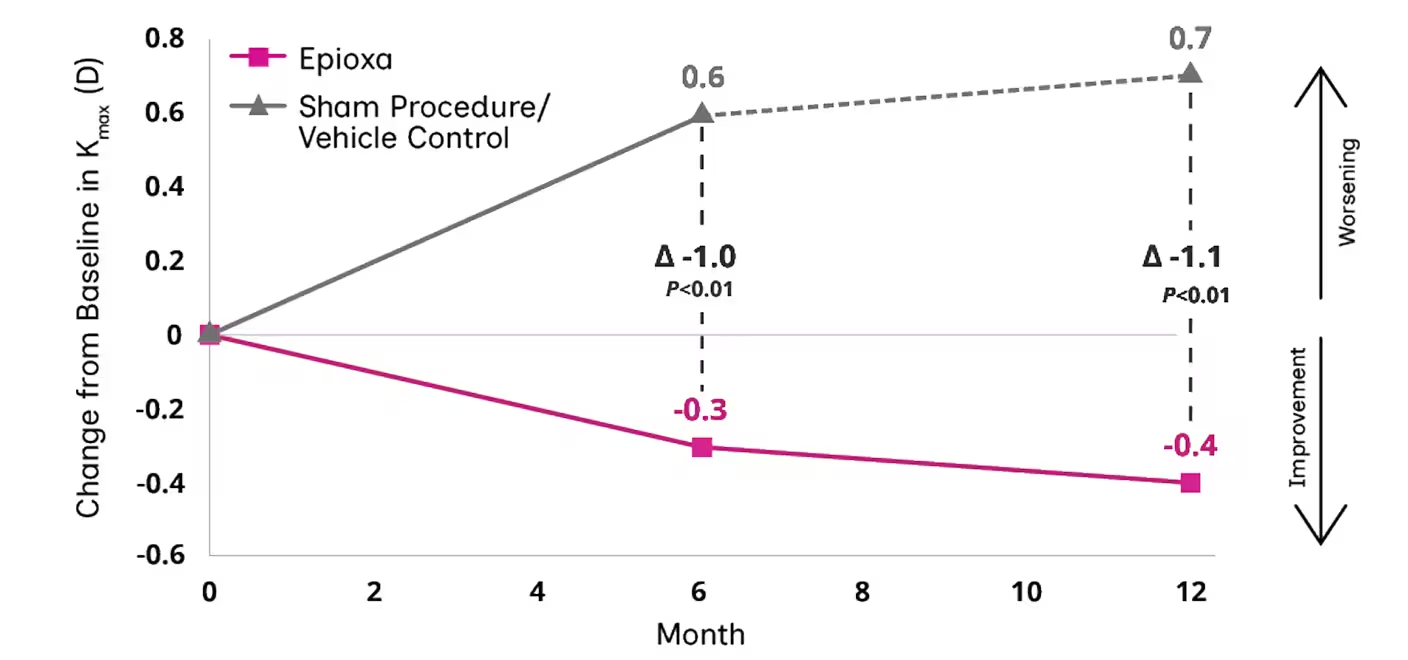
Study 1: LS Mean Change from Baseline in Kmax
Study 2: LS Mean Change from Baseline in Kmax
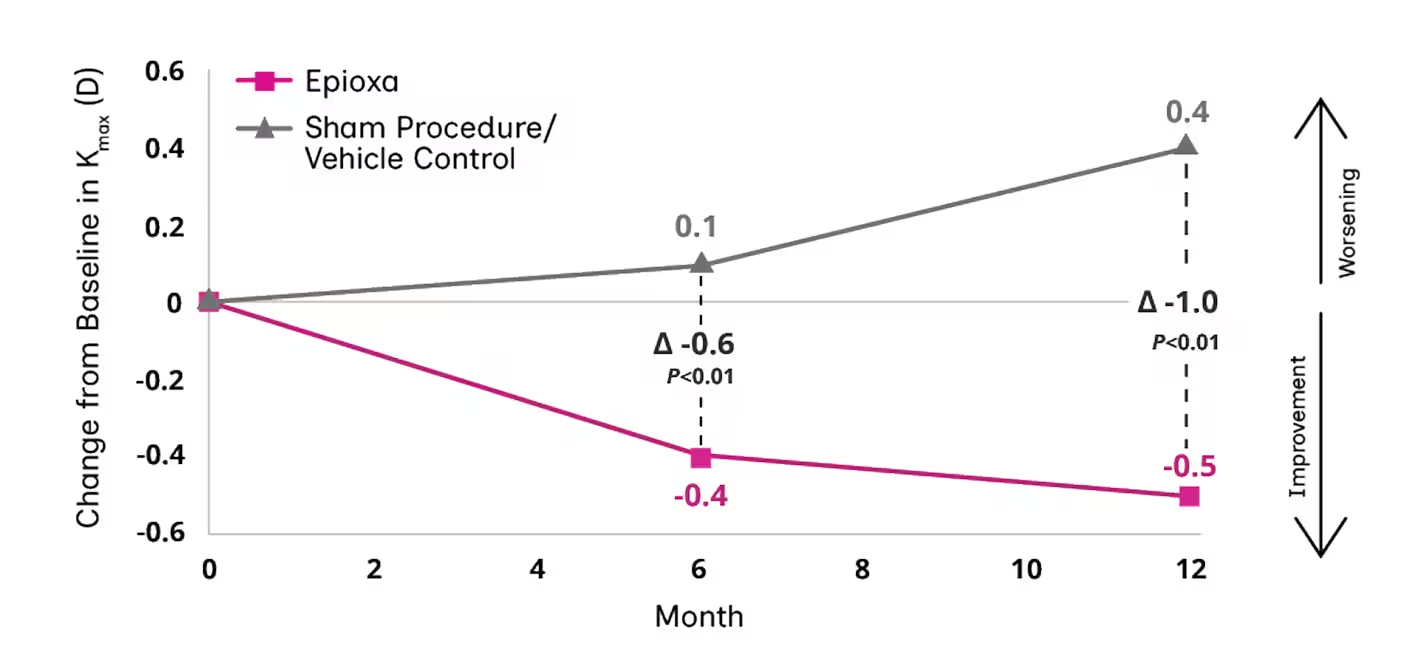
At Months 6 and 12, mean change from baseline was Kmax or -1.0 D in Study 1 (P<0.01) and Study 2 (P=0.01), respectively1
Epioxa CXL therapy was proven in 2 clinical studies to enhance corneal stability1
-
Epioxa Study 1 Design1,2*
279 eyes in participants aged between 13 and 53 years were randomized in 2:1 ratio into CXL with Epioxa and sham procedure/vehicle control arms. The primary efficacy endpoint was change from baseline in maximum corneal curvature (Kmax) ≥1.0 D at Month 6 (P<.05). Sham procedure/vehicle control eyes could cross over and receive CXL with Epioxa at Month 6 and were followed to Month 12. 19% of eyes (54) had KC severity grade moderate or severe. All participants showed topographic and clinical evidence of KC.
-
Epioxa Study 2 Design1-3*
312 eyes in participants aged between 13 and 51 years were randomized in 2:1 ratio into CXL with Epioxa and sham procedure/vehicle control arms. The primary efficacy endpoint was the change from baseline in Kmax of ≥1.0 D at Month 12 (P<.05). 22% of eyes (70) were aged ≥30 years and 14% of eyes (45) had KC severity grade moderate or severe. All participants showed topographic and clinical evidence of KC.
*Epioxa treatment safety and efficacy were studied in 2 prospective, multicenter, randomized, sham procedure/vehicle control trials.1
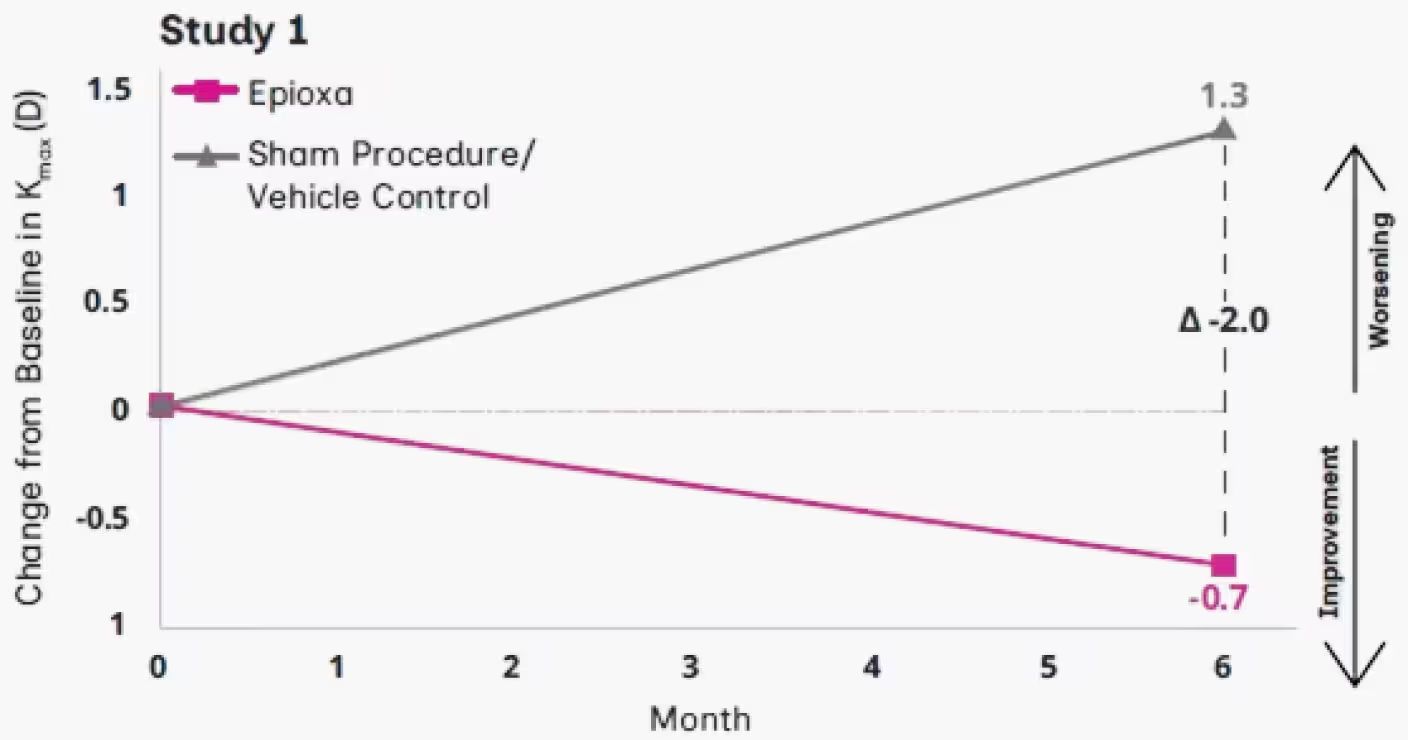
In Study 1, the treatment effect was nearly double in patients aged 13-281
At Month 6, mean change from baseline in Kmax -2.0 D in Study 11†
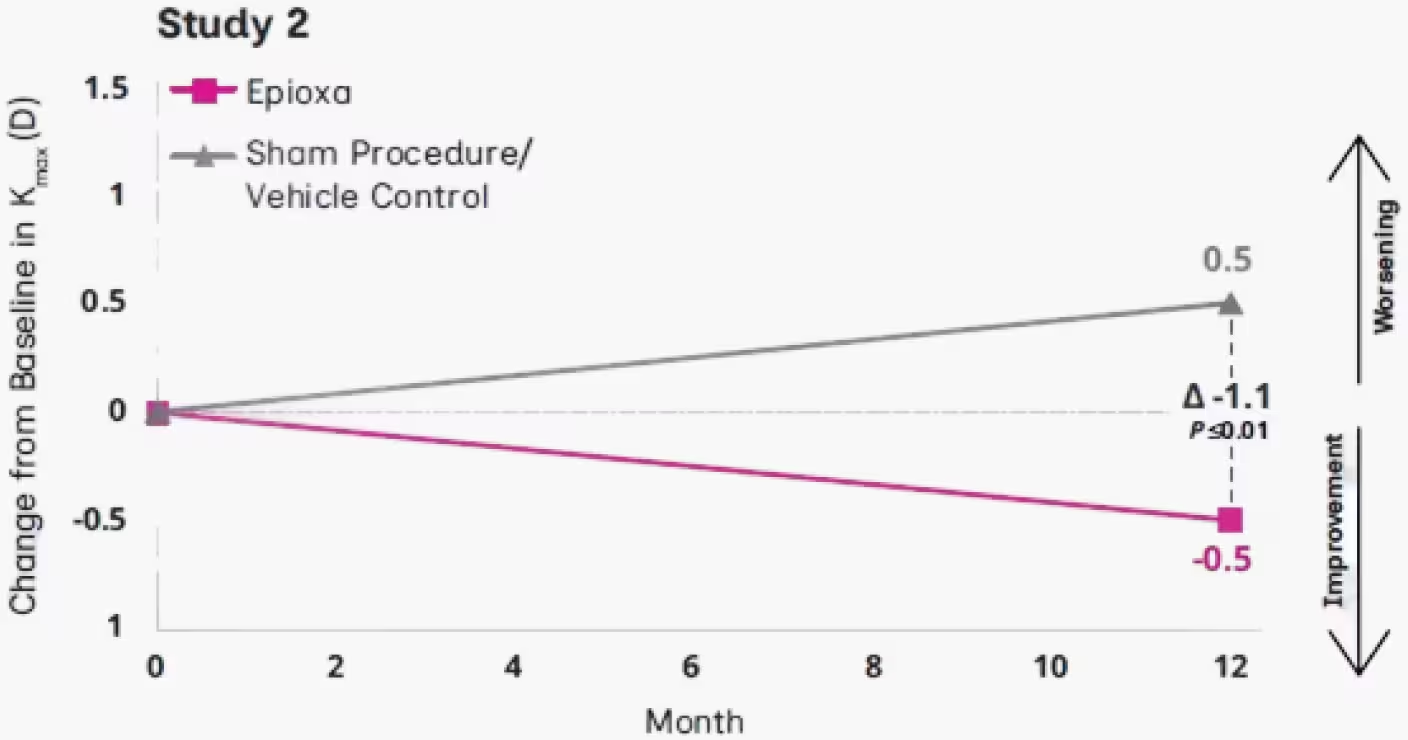
In Study 2, younger patients benefited from early Epioxa treatment
At Month 12, mean change from baseline in Kmax was 1.1 D in Study 2 (P≤0.01)1,2‡
†For patients <29 years of age.1
‡For patients <30 years of age.1
Safety data for Epioxa
| THE MOST COMMON ADVERSE REACTION OCCURRED IN 31% OF CXL-TREATED EYES1 | ||
|---|---|---|
| Conjunctival hyperaemia | ||
| ADVERSE REACTIONS OCCURRED IN 5% TO 25% OF CXL-TREATED EYES1 | ||
| Corneal opacity (haze) | Punctate keratitis | Eye Pain |
| Eye irritation | Lacrimation increased | Corneal epithelium defect |
| Eyelid edema | Corneal striae | Visual acuity reduced |
| Dry eye | Anterior chamber flare | Photophobia |
Patients were able to get their second Epioxa treatment as soon as 1 week later1
Patients may pay as little as $0 for both Epioxa and the cross-linking procedure§
Explore access and support options§Epioxa copay program is available for commercially insured eligible patients only. Additional restrictions may apply. Subject to change; for full terms and conditions from EpioxaCareConnect.
CXL=cross-linking; D=diopter; Kmax=maximum corneal curvature.
References:
- EPIOXA HD and EPIOXA Prescribing Information. Glaukos Corporation; 2025.
- Data on File.
- Smith VL, et al. Poster presented at Association for Vision and Ophthalmology; May 4-8 2025; Salt Lake City, UT.
IMPORTANT SAFETY INFORMATION
Contraindications
EPIOXA™ HD and EPIOXA™ are contraindicated in patients with known hypersensitivity to benzalkonium chloride (BAC) or any ingredients in EPIOXA HD and EPIOXA. Epithelium-on corneal collagen cross-linking is contraindicated in aphakic and pseudophakic patients without a UV-blocking intraocular lens.
Warnings and Precautions
Corneal collagen cross-linking should be used with caution in patients with a history of herpetic keratitis due to the potential for reactivation of herpes keratitis.
Adverse Reactions
The most common adverse reaction was conjunctival hyperaemia (31%). Other adverse reactions, occurring in 5% to 25% of eyes included: corneal opacity (haze), photophobia, punctate keratitis, eye pain, eye irritation, increased lacrimation, corneal epithelium defect, eyelid oedema, corneal striae, visual acuity reduced, dry eye, and anterior chamber flare.
Dosage and Administration
EPIOXA HD and EPIOXA are for topical ophthalmic use. NOT for injection or intraocular use.
EPIOXA HD and EPIOXA are supplied in single-dose syringes. Discard opened syringes after use.
EPIOXA HD and EPIOXA are for use with the O2n System and Boost Goggles only.
Refer to the O2n System Operator’s Manual and Boost Goggles User Guide for device instructions.
INDICATIONS AND USAGE
EPIOXA™ HD (riboflavin 5’-phosphate ophthalmic solution) 0.239% and EPIOXA™ (riboflavin 5’-phosphate ophthalmic solution) 0.177% are photoenhancers indicated for use in epithelium-on corneal collagen cross-linking for the treatment of keratoconus in adults and pediatric patients aged 13 years and older, in conjunction with the O2n™ System and the Boost Goggles®.
Please see full Prescribing Information for EPIOXA HD and EPIOXA at www.Epioxa.com.
You are encouraged to report all side effects to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.
You may also call Glaukos at 1-888-404-1644.